The relationship between (EMF) & the temperature on Daniel's cell solutions.
In this article we will discuss the relationship between electromotive
force (EMF) and the temperature in Daniel's cell solutions.
In other words: Why does the voltage decrease with increasing
temperature?
Prerequisites
- Be familiar with the working mechanism of the galvanic cell
- How a balanced equation works
- The oxidation-reduction reaction
- How exothermy affects the reaction
Why does the voltage decrease with increasing temperature?
It is common to think about the relationship between E(cell) and
temperature, since it is an experiment in the physical chemistry
Laboratory.
So what happens in this experiment and why?
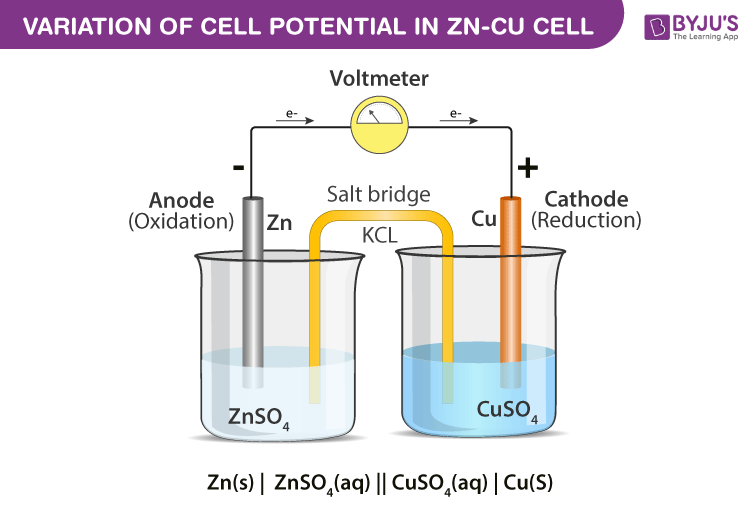
In this experiment you have to prepare Daniel's cell (galvanic cell), and this is quite simple: you take two beakers, a cathode and anode, and in our cell it will be (copper and zinc) so now put them into the beakers, where we have already put ions solutions (CuSo4, ZnSo4), (Cu+2, Zn+2) and a salt bridge containing (KCL) between the beakers, then connect the cathode and the anode with a wire then place a galvanometer on the wire to measure the electromotive force (EMF) the movement of electrons between cathode and anode or the E(cell) between the cathode and the anode, while placing the solutions beakers in a thermostat that raises the temperature of the cell solutions so it will be warmed over time to see what happens to the E(cell) values.
Daniel’s cell ( galvanic cell ):
After the experimental measurements on our cell, we can plot them in the diagram and we get these results:
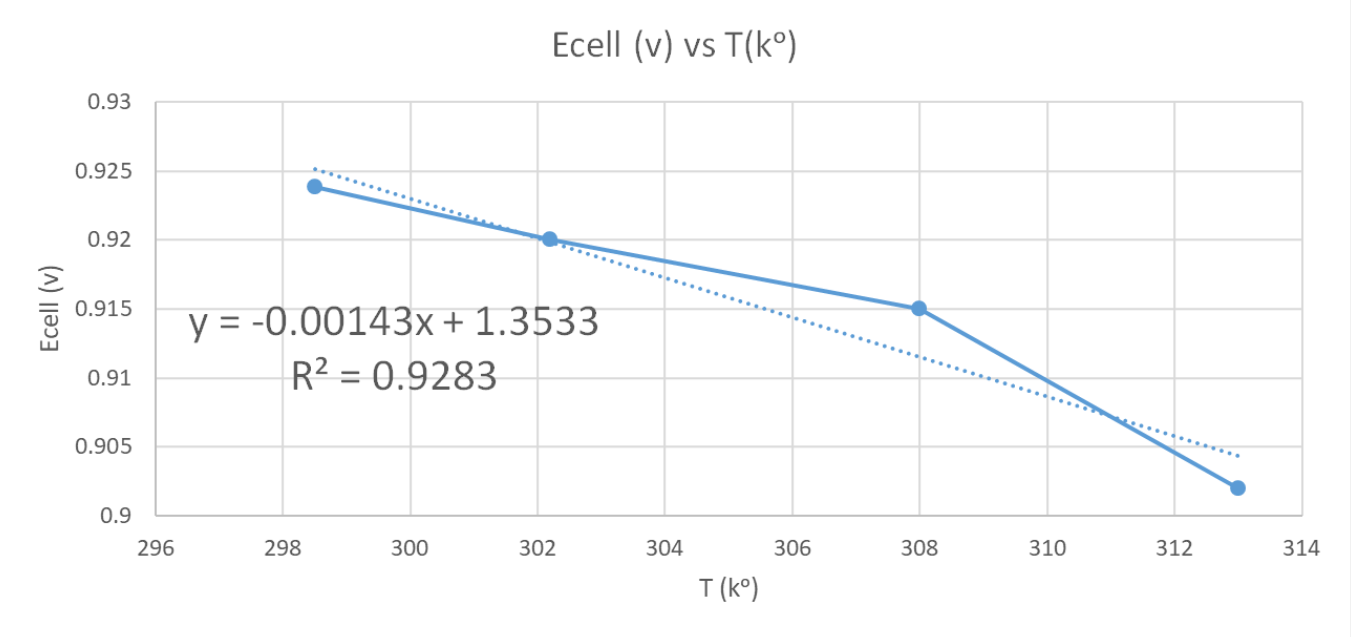
As we can see in the diagram, the slope in the equation has a negative value, so now we know that it is an inverse relationship between E(cell) and T(kᵒ).
So now we know how to build a galvanic cell and what kind of relationships between E(cell) and T (kᵒ). Now we ask ourselves, why is the relationship inverse?
First of all, from the experimental values we can calculate the Gibbs
free energy and enthalpy from the experimental values using these
equations: Gibbs free energy: ∆G=-n F E
Where:
n: Number of moles
F: Faraday’s constant
E: Electromotive force
Enthalpy: ∆H = -nFE-nFT (-∆E/∆T) Slope of diagram = ∆E/∆T
In the end, we will raise the temperature and get a negative values for both of them and now we should know that our reaction is (spontaneous and exothermic), so that as the temperature increases, our reaction will move towards the reactant (shift to left ) in our oxidation-reduction reaction, so that the reaction will not proceed well and the electromotive force will be reduced in our galvanic cell.
Now let’s see the reason why it’s an inverse relation between EMF and T(kᵒ) at the ionic or atomic level and you can say that the reason is:
In our galvanic cell, we have zinc and copper connected by a wire, so the electrons move from the anode (zinc) to the cathode (copper) because copper attracts the electrons more than zinc, so it takes electrons from zinc through the wire between them, and that's the voltage of cell (movement of electrons), so when electrons go to the copper metal, the free copper ions (Cu+2) actually in our copper sulfate solution, so we have a free copper ion in our solution, and when these electrons go to the copper metal, it gives them to the copper ions from It’s solution and becomes neutral, so it sticks to the copper metal and becomes neutral so it increases the size of the metal (notice the difference in size of the cathode between the beginning and the ending of the experiment), and this happens at room temperature (normal case), but when we increase the temperature of the cell solutions and we know that our reaction is spontaneous and exothermic, as can be seen from the experimental values of Gibbs-free energy and enthalpy (negative values), then we now know that the energy coming from the temperature goes to the ions in the solution and they gain a spontaneous kinetic energy and start banging against the walls of the container, away from the metal, so our ions become two parts, one that gains energy and moves away from the metal, so it is freed from its main task (taking electrons from the metal and becoming a neutral) and does not take electrons from the metal, And the other part goes to the metal and work fine, That’s why it continues the movement of electron in the wire, So as the temperature increases, the number of ions moving away from the metal increases too and the use of electrons pulled from the zinc to the copper metal decreases as the increasing of copper ions that’s freed from its main task, So the copper metal becomes electron rich because there’s no large number of ions as the begging of the reaction that takes electrons from the copper metal so there will be decreasing of electrons that were pulled from zinc and the electron pull decreases so the movement of electrons in the wire decreases and the voltage decreases as the temperature increases because the voltage E(cell) is the electron movement in the wire.
Visit this additional link to view the synthesis and retrosynthesis file of Atorvastatin
And here are a few of my lab reports at university: